
AskSia
2. Inversion of sucrose is a bimolecular reaction of sucrose with water, producing glucose and fructose:
a) What is the reaction order if the reaction takes place in dilute aqueous solution (great excess of water)?
The respective half-lifes of the reaction in dilute and acidified aqueous solutions were measured by polarimetry at various temperatures, with the initial concentration of sucrose , and it was found: , .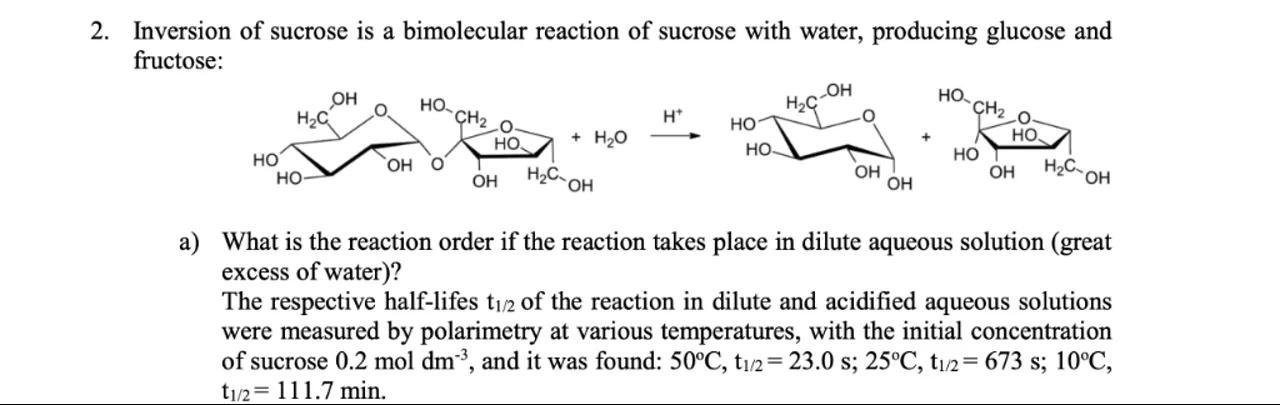

Answer from Sia
Posted about 2 years ago
Solution
1
Determine the reaction order: The reaction order can be determined by analyzing the reaction conditions and the given data. In a dilute aqueous solution, the concentration of water is much greater than that of sucrose and remains nearly constant throughout the reaction. Therefore, the reaction rate is independent of the concentration of water
2
Apply the definition of reaction order: Since the concentration of water does not change appreciably and does not affect the rate, the reaction order with respect to water is zero. The reaction is catalyzed by a hydrogen ion, but the catalyst does not affect the overall reaction order. Thus, the reaction order is determined solely by the concentration of sucrose
3
Use the half-life data: The half-life of a reaction is the time required for the concentration of a reactant to decrease to half its initial value. For a first-order reaction, the half-life is independent of the initial concentration. The provided half-life data does not depend on the initial concentration of sucrose, which suggests that the reaction is first-order with respect to sucrose
Answer
The reaction order is first-order with respect to sucrose.
Key Concept
Reaction order determination in dilute solutions
Explanation
In dilute solutions where the solvent's concentration remains constant, the reaction order is determined by the reactant whose concentration changes and affects the rate, which in this case is sucrose. The constant half-life regardless of initial concentration indicates a first-order reaction with respect to sucrose.
Not the question you are looking for? Ask here!
Enter question by text
Enter question by image
Unlock Smarter Learning with AskSia Super!
Join Super, our all-in-one AI solution that can greatly improve your learning efficiency.
30% higher accuracy than GPT-4o
Entire learning journey support
The most student-friendly features
Study Other Question