
AskSia
5)
Hydrogen can be produced by the steam reforming of methane as follows:
a) If is at , what is the value of ?
b) If is at , what is the value of ?
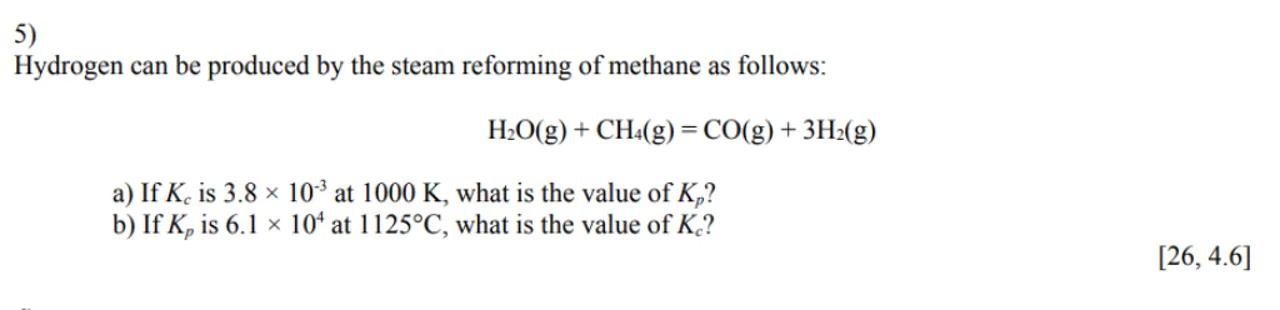

Answer from Sia
Posted about 2 years ago
Solution
1
Identify the relationship between $K_p$ and $K_c$: The relationship between the equilibrium constants and is given by the equation , where is the ideal gas constant (0.0821 L·atm/mol·K), is the temperature in Kelvin, and is the change in the number of moles of gas in the reaction
2
Calculate $\Delta n$ for the reaction: For the given reaction,
3
Calculate $K_p$ using $K_c$: Given at K, we use the equation from step 1 to find
a Answer
Key Concept
The relationship between and involves the temperature and the change in the number of moles of gas.
Explanation
is calculated from by considering the change in moles of gas and the temperature in Kelvin.
1
Identify the relationship between $K_p$ and $K_c$ for part b: The relationship is the same as in part a,
2
Convert temperature to Kelvin: The temperature is given as 1125°C, which is 1125 + 273.15 = 1398.15 K
3
Calculate $K_c$ using $K_p$: Given , we rearrange the equation from step 1 to find
b Answer
Key Concept
The relationship between and is used to calculate one from the other, given the temperature and the change in moles of gas.
Explanation
is calculated from by using the ideal gas constant and the temperature in Kelvin, along with the change in moles of gas.
Not the question you are looking for? Ask here!
Enter question by text
Enter question by image
Unlock Smarter Learning with AskSia Super!
Join Super, our all-in-one AI solution that can greatly improve your learning efficiency.
30% higher accuracy than GPT-4o
Entire learning journey support
The most student-friendly features
Study Other Question
